Identification of mutations in laboratory-evolved microbes from next-generation sequencing data using breseq.
#Pbp3 is essential manual
Molecular Cloning: A Laboratory Manual 2nd edn (Cold Spring Harbor Laboratory Press, 1989).ĭeatherage, D. Fluorescent reporters for studies of cellular localization of proteins in Staphylococcus aureus. Isolation and characterization of a Tn 551-autolysis mutant of Staphylococcus aureus. Inactivation of the SauI type I restriction-modification system is not sufficient to generate Staphylococcus aureus strains capable of efficiently accepting foreign DNA. Molecular coordination of Staphylococcus aureus cell division. A DNA damage-induced, SOS-independent checkpoint regulates cell division in Caulobacter crescentus. FtsEX acts on FtsA to regulate divisome assembly and activity. Beta-lactam antibiotics induce a lethal malfunctioning of the bacterial cell wall synthesis machinery. A central role for PBP2 in the activation of peptidoglycan polymerization by the bacterial cell elongation machinery. The tubulin homologue FtsZ contributes to cell elongation by guiding cell wall precursor synthesis in Caulobacter crescentus. EzrA contributes to the regulation of cell size in Staphylococcus aureus. Identification and characterization of a negative regulator of FtsZ ring formation in Bacillus subtilis. In situ probing of newly synthesized peptidoglycan in live bacteria with fluorescent d-amino acids. Imaging protein molecules using FRET and FLIM microscopy. Peptidoglycan synthesis drives an FtsZ-treadmilling-independent step of cytokinesis. Functional redundancy of division specific penicillin-binding proteins in Bacillus subtilis.

Evidence for a dual role of PBP1 in the cell division and cell separation of Staphylococcus aureus. Cloning, characterization, and inactivation of the gene pbpC, encoding penicillin-binding protein 3 of Staphylococcus aureus. Role of PBP1 in cell division of Staphylococcus aureus. Cell shape dynamics during the staphylococcal cell cycle. Bacterial cell wall synthesis: new insights from localization studies. Bacterial cell wall biogenesis is mediated by SEDS and PBP polymerase families functioning semi-autonomously.
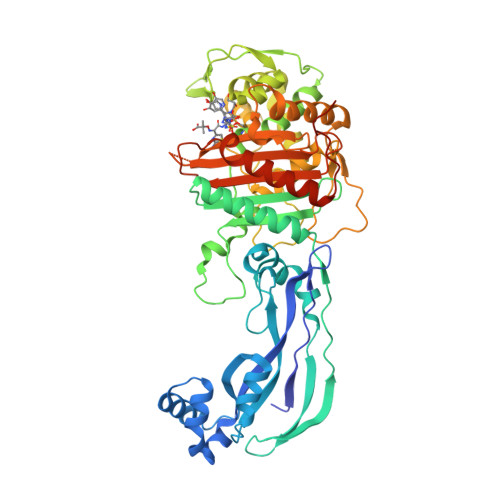
SEDS proteins are a widespread family of bacterial cell wall polymerases. RodA as the missing glycosyltransferase in Bacillus subtilis and antibiotic discovery for the peptidoglycan polymerase pathway. FtsW is a peptidoglycan polymerase that is functional only in complex with its cognate penicillin-binding protein. From the regulation of peptidoglycan synthesis to bacterial growth and morphology. Movement dynamics of divisome proteins and PBP2x:FtsW in cells of Streptococcus pneumoniae.
#Pbp3 is essential how to
How to get (a)round: mechanisms controlling growth and division of coccoid bacteria. From models to pathogens: how much have we learned about Streptococcus pneumoniae cell division? Environ. The physiology of bacterial cell division. The bacterial actin MreB rotates, and rotation depends on cell-wall assembly. Coupled, circumferential motions of the cell wall synthesis machinery and MreB filaments in B. Processive movement of MreB-associated cell wall biosynthetic complexes in bacteria. The actin-like MreB proteins in Bacillus subtilis: a new turn. Regulation of bacterial cell wall growth. We conclude that RodA–PBP3 and FtsW–PBP1 mediate sidewall and septal PGN incorporation, respectively, and that their activity must be balanced to maintain coccoid morphology.Įgan, A. In the absence of these proteins, the divisome appears as multiple rings or arcs that drive lateral PGN incorporation, leading to cell elongation. We propose that the FtsW–PBP1 pair has a role in stabilizing the divisome at midcell. Although PBP1 is an essential protein, a mutant lacking PBP1 transpeptidase activity is viable, showing that this protein has a second function. A lack of RodA–PBP3 resulted in more spherical cells due to deficient sidewall PGN synthesis, whereas depletion of FtsW–PBP1 arrested normal septal PGN incorporation. aureus RodA–PBP3 and FtsW–PBP1 probably constitute cognate pairs of interacting proteins.

Here, we elucidate why coccoid bacteria, such as Staphylococcus aureus, also possess two SEDS–bPBP pairs. Rod-shaped bacteria have two SEDS–bPBP pairs, involved in elongation and division. These include two key protein families, SEDS transglycosylases and bPBP transpeptidases, proposed to function in cognate pairs. Bacteria maintain cell shape by directing PGN incorporation to distinct regions of the cell, namely, through the localization of late-stage PGN synthesis proteins. Peptidoglycan (PGN) is the major component of the bacterial cell wall, a structure that is essential for the physical integrity and shape of the cell.
